無料セッションのポータルサイトからご参加ください!
https://link.cancerx.jp/wcw2022-opensession-portal
【COVID-19 パンデミック下でのファイザーとモデルナのワクチンの迅速承認プロセスから学べることは?】What we can learn from the Pfizer and Moderna vaccine rapid approval process under the COVID-19 pandemic(English below)
World Cancer Week 2022 最終日、明朝1つ目のセッションでは3名の豪華なスピーカーご登壇いただき ”がんの治療法の承認プロセスをどうやって加速させることができるか?” について話し合うセッションを行います。
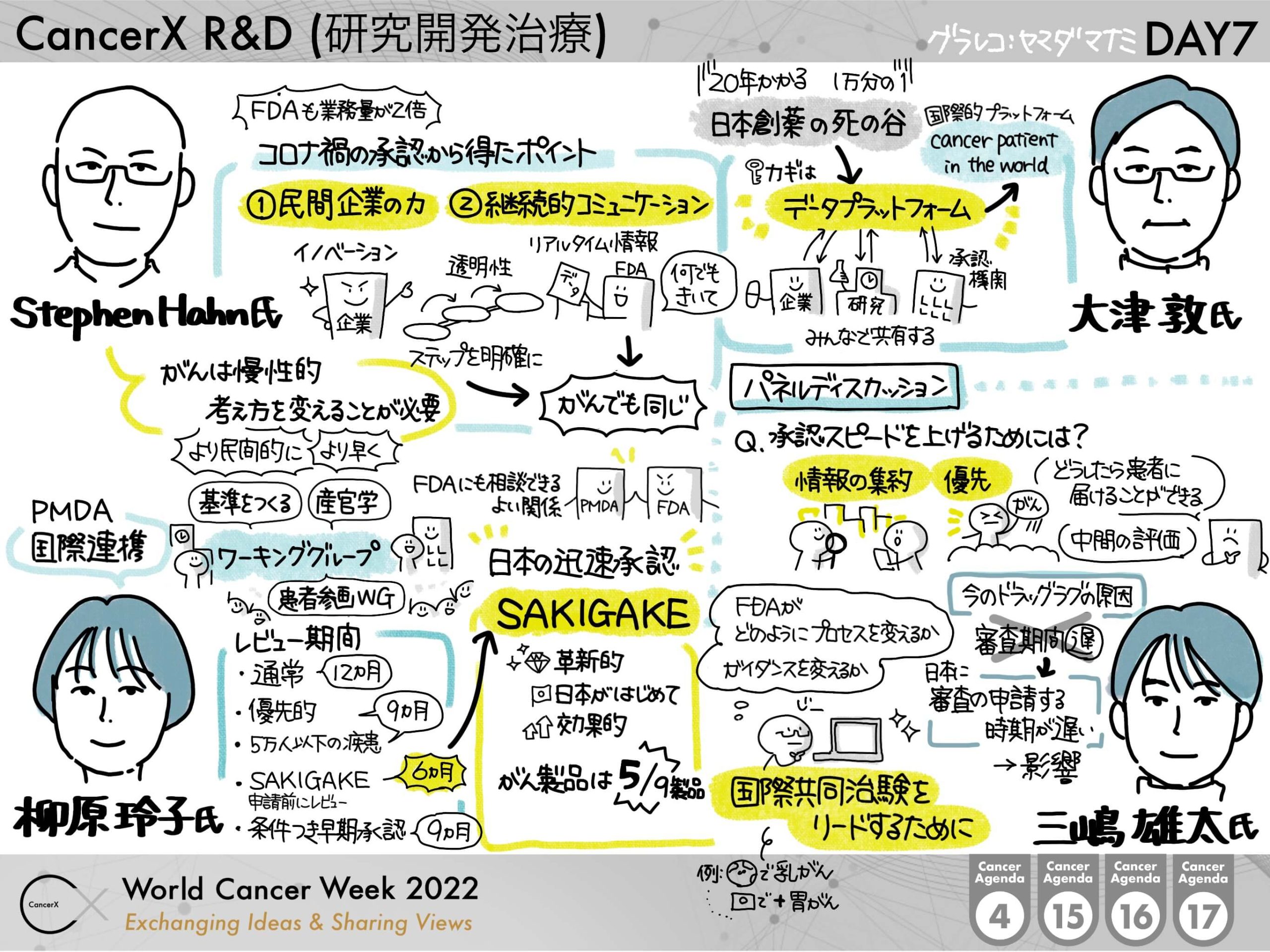
視聴リンク:https://link.cancerx.jp/wcw2022-021-randd
Hahnさんは、トランプ政権下で2019-2021の間に FDAの長官を務めた方で、COVID-19のパンデミック下、ファイザーとモデルナの迅速承認において指揮をとった方です。もともと Oncologist というバックグラウンドから、抗がん剤で経験した臨床試験や承認プロセスは、今回の迅速承認に活きていると事前ミーティングでは伺っています。今回は、この迅速承認ががんの領域の迅速承認プロセスに与える学びはなにか?ということも引き出せればと思っております。
柳原さんは、日本の審査機関である PMDA の審査第五部の審査役をつとめており、審査第五部というのはがん治療薬の審査を担当している部門になります。Hahn さんのお話と併せて、日米の審査機関の貴重なお話とコミュニケーションが聞けると思います。また、COVID-19のワクチンの審査プロセスや、ドラッグラグの現状など審査役の方から直接お話を聞ける貴重な機会になると思います。
大津先生は国立がんセンター東病院の病院長で、年間9,000人を超える新患の方が訪れる国内トップクラスのがん専門病院の長として、なぜ新薬の創出には時間がかかるのか、日本における臨床試験の課題や併設する先端医療開発センター(EPOC)を通してお話し頂く予定です。
国際対がん連合(UICC)とも連携しているため、無料公開の英語のセッションになります。同時通訳付きですのでもしよかったら聞いてみてください。
(CancerX 三嶋)
Today is World Cancer Day and the last day of World Cancer Week 2022, organized by CancerX. The first session of the morning is a discussion on “How can we accelerate the approval process for cancer treatments?
Dr. Hahn was the FDA Commissioner from 2019-2021 under the Trump Administration and led the rapid approval of Pfizer and Moderna during the COVID-19 pandemic. I was told at the pre-meeting that his background as an oncologist and his experience with clinical trials and the approval process for cancer drugs was very beneficial in the rapid approval process. In this session, I would like to ask what lessons can be learned from this rapid approval process for the oncology field?
Dr. Yanagihara is a reviewer at the PMDA, the Japanese regulatory agency, and comes from the office of V that is responsible for the review of oncology drugs. Together with Dr. Hahn’s talk, we will be able to hear valuable stories and communication from the reviewing bodies in the US and Japan. It will be a valuable opportunity for the audience to hear directly from the reviewers about the review process for the COVID-19 vaccine and the current status of the drug lag in Japan.
Dr. Ohtsu is the Director of the National Cancer Center East Hospital, one of the top cancer hospitals in Japan with over 9,000 new patients a year, and will talk about why it takes so long to create new cancer drugs, the challenges of clinical trials in Japan, and the Advanced Medical Development Center (EPOC) which is attached to the hospital.
The session will be free and open to the public in English, in connection with the International Union Against Cancer (UICC).
Simultaneous interpretation will be available. The archive will be available later here.
https://link.cancerx.jp/wcw2022-opensession-portal
(Yuta Mishima, CancerX)